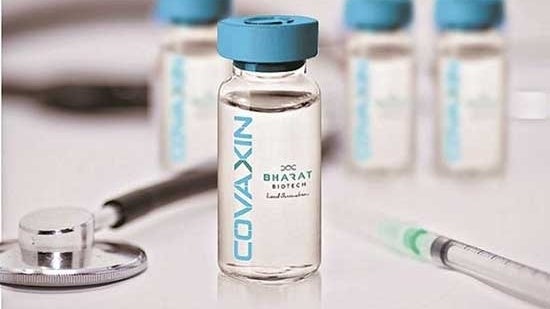
The US company said in a regulatory filing that Bharat Biotech’s partner for USA and Canada for COVID-19 vaccine Covaxin has initiated a rolling submission to Health Canada for the vaccination.
The move follows the release by Bharat Biotech of Phase 3 clinical trial results, which demonstrated efficacy and safety in nearly 25,800 adults, it said on Thursday.
As information continues to come in to accelerate the overall review process, this allows Health Canada to start its review right away.
Ocugen initiated the rolling submission through its affiliate, Vaccigen, Ltd. Health Canada will make a decision upon review of the evidence submitted that supports its safety, efficacy and quality.
“We thank Health Canada for their upcoming review of Covaxin and look forward to working with them so that we can offer the possibility of another safe and effective option to be used in their fight against COVID-19 and its Delta variant,” Dr. Shankar Musunuri, Chairman of the Board, Chief Executive Officer and Co-Founder of Ocugen said.
Ocugen said, the rolling submission process was recommended and accepted under the Minister of Health’s Interim Order Respecting the Importation, Sale and Advertising of Drugs for Use in Relation to COVID-19 and transitioned to a new drug.
Covaxin has been developed in collaboration with the Indian Council of Medical Research (ICMR) National Institute of Virology.